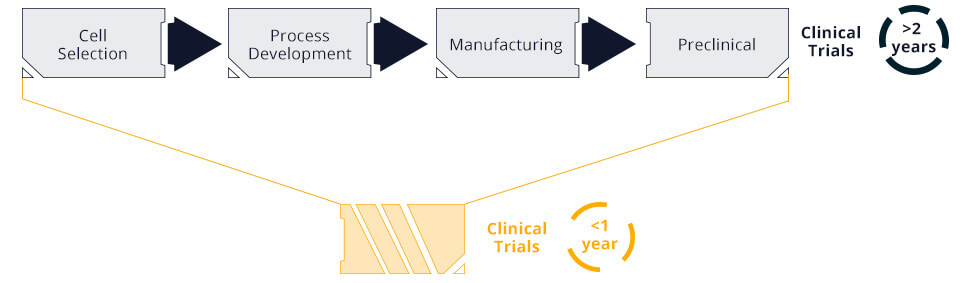
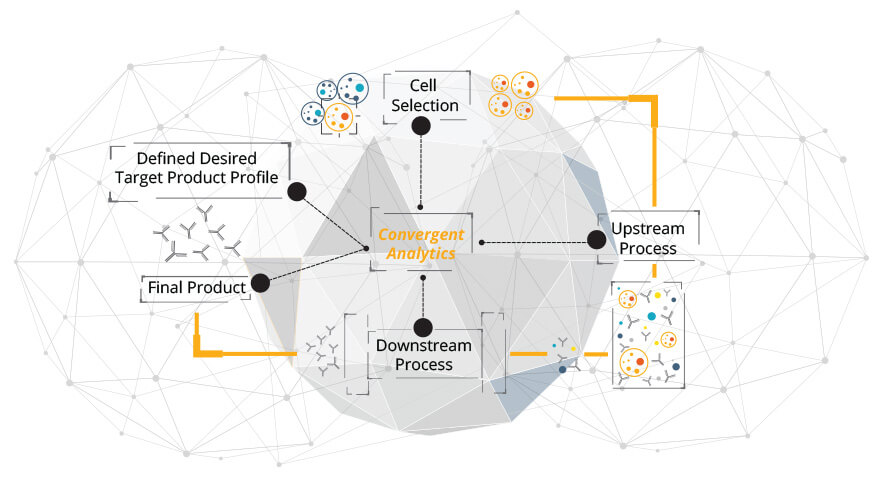
Tychan is a clinical-stage biotechnology company focused on bringing life-saving treatments to those in need through disruptive technologies. We seek to reduce the development timeline of therapeutics leading to clinical application - a major unmet need in the context of infectious diseases. Our technology platform integrates convergent analytics with innovative manufacturing processes to enable rapid production of safe and effective biologics for human trials.
The name Tychan (pronounced Tyk-AN), is derived from Tyche, the Greek goddess who governs the fortune and prosperity of a city and 安 (ān), meaning peace in Mandarin.
We believe this integrated approach will enable Singapore, and the world, to respond to emerging infections during and not after epidemics.
Ram Sasisekharan is the Alfred H. Caspary Professor of Biological Engineering and Health Sciences and Technology in the Department of Biological Engineering at Massachusetts Institute of Technology (MIT), as well as a member of MIT’s Koch Institute for Integrative Cancer Research. He has founded multiple biotechnology companies including Momenta Pharmaceuticals, Cerulean Pharma and Visterra Pharmaceuticals. Ram has also won numerous prestigious awards throughout his scientific career. In 2017, he was awarded the Agilent Thought Leader Award in recognition of his contributions in the field of biologics characterization. He received his B.S. in Physical Sciences from Bangalore University, his M.S. in Biophysics from Harvard University and his Ph.D. in Medical Sciences from Harvard Medical School.
Professor Ooi Eng Eong, a clinician scientist, is currently the deputy director of Emerging Infectious Diseases Programme at Duke-National University of Singapore Medical School. He is also the co-director of Viral Research And Experimental Medicine Centre @ SingHealth Duke-NUS (ViREMiCS). Eng Eong has published numerous works in high-impact journals and is a winner of numerous prestigious awards throughout his scientific career.
He received his M.B.B.S. from the University of Nottingham and his Ph.D. from the National University of Singapore.
- Randall E. Woods (CEO, Member of the Board)
- Teo Ming Kian (Chairman)
- Professor Ram Sasisekharan (Founder)
- Professor Ooi Eng Eong (Founder)
- Syed Fidah bin Ismail Alsagoff (Member)
Appointed as CEO and Board member in 2021, Randall Woods is an accomplished life science entrepreneur as well as a pharmaceutical and biotechnology executive.
He previously served as the CEO of Sophiris Bio; President and CEO at NovaCardia, Inc. prior to its acquisition by Merck & Co., and as CEO of Corvas International, Inc., acquired by Dendreon. Prior to Corvas, Mr. Woods was President of Boehringer Mannheim’s US Pharmaceutical operations and spent over 20 years at Eli Lilly & Company.
Mr. Woods has also served as the Chairman of the Advisory Board of the University of California, San Diego’s Sulpizio Family Cardiovascular Center and as Chairman of the Board of Directors for BIOCOM, a life science industry association in California.
Mr Teo Ming Kian has been Chairman of Tychan Pte Ltd since April 2016.
He is concurrently Chairman of Vertex Venture Holdings Ltd; Tessa Therapeutics Pte Ltd; Temasek Foundation Ecosperity CLG Ltd; Temasek Life Sciences Laboratory Ltd (TLL), Temasek Life Sciences Ventures Private Limited and Temasek Lifesciences Accelerator Pte Ltd - TLL’s subsidiary and joint venture respectively.
Mr Teo is also a Board Director of Temasek Holdings and Interel Pte Ltd, as well as Fellow of the Singapore Academy of Engineering.
Before his retirement from the Singapore Civil Service, Mr Teo held Permanent Secretary and Executive Chairman positions in several Singapore government ministries and agencies.
Mr Teo was conferred the Singapore Public Administration Medal (Gold) in 1993, the Commander First Class — Royal Order of the Polar Star (Sweden) in 1994, the Distinguished Alumni Award, Monash University, Australia in 1999, the Meritorious Service Medal in 2008 and the Defence Technology Medal (Distinguished Leadership) in 2015.
Mr Teo holds a Bachelor of Engineering (First Class Honours) degree in Mechanical Engineering from Monash University in Australia, and Master of Science degree in Management Studies from MIT (Massachusetts Institute of Technology).
Ram Sasisekharan is the Alfred H. Caspary Professor of Biological Engineering and Health Sciences and Technology in the Department of Biological Engineering at Massachusetts Institute of Technology (MIT), as well as a member of MIT’s Koch Institute for Integrative Cancer Research. He has founded multiple biotechnology companies including Momenta Pharmaceuticals, Cerulean Pharma and Visterra Pharmaceuticals. Ram has also won numerous prestigious awards throughout his scientific career. In 2017, he was awarded the Agilent Thought Leader Award in recognition of his contributions in the field of biologics characterization. He received his B.S. in Physical Sciences from Bangalore University, his M.S. in Biophysics from Harvard University and his Ph.D. in Medical Sciences from Harvard Medical School.
Professor Ooi Eng Eong, a clinician scientist, is currently the deputy director of Emerging Infectious Diseases Programme at Duke-National University of Singapore Medical School. He is also the co-director of Viral Research And Experimental Medicine Centre @ SingHealth Duke-NUS (ViREMiCS). Eng Eong has published numerous works in high-impact journals and is a winner of numerous prestigious awards throughout his scientific career.
He received his M.B.B.S. from the University of Nottingham and his Ph.D. from the National University of Singapore.
Dr Fidah Alsagoff is a Senior Managing Director at Temasek International Pte Ltd, where he has been since 2008. He is currently Head, Life Sciences. In addition, he is also Executive Director of Sheares Healthcare Management Pte Ltd and serves on the Board of Trustees of the Singapore University of Technology Design. His career includes nearly a decade in social entrepreneurship where he started up several programs and enterprises catering to hitherto unmet healthcare needs of patients, with the latest enterprise being the establishment of the Singapore Cord Blood Bank.
Fidah graduated from the National University of Singapore with a Bachelors of Medicine and Surgery, and later with a Masters of Medicine in Public Health. He attended INSEAD on a Lee Kuan Yew Scholarship for post-graduate studies, and subsequently graduated with an Executive MBA (with distinction) and valedictorian of his class.